Epilepsy is a “short circuit in the brain”. The first successful surgery for uncontrolled epilepsy was performed by Sir Victor Horsley, on 25th May, 1886
Epilepsy is a condition in which a person has recurrent seizures. A seizure is defined as an abnormal, disorderly discharging of the brain’s nerve cells, resulting in a temporary disturbance of motor, sensory, or mental function. Epilepsy is like a short circuit in the brain, just as the short circuit in the house fuses off all the electrical appliances, the short circuit in the brain fuses off brain and the brain stops functioning for few minutes.
There are many types of seizures, depending primarily on what part of the brain is involved. The term epilepsy says nothing about the type of seizure or cause of the seizure, only that the seizures happen again and again. A stricter definition of the term requires that the seizures have no known underlying cause. This may also be called primary or idiopathic epilepsy. Episodes of abnormal electrical activity within the brain result in seizures. The specific area of the brain affected by the abnormal electrical activity may result in a particular type of seizure.
Epilepsy is a fairly common disease. WHO data suggests that 1 in 20 persons may have an epileptic seizure in his/ her lifetime and at least 1in 200 goes on to develop epilepsy. [1] Anticonvulsant drug therapy using one or more drugs works as an effective tool to suppress seizures in only 70% of the patients; the remaining 30% are either not responsive or suffer major side effects. [2] Surgical resection then forms the next line of management in selected patients. However, in some cases, surgical resection may not be possible; hence arises the need for alternative therapies.
Healthy people may have seizures under certain circumstances. Most of the seizures causes are unknown under which circumstance it is called primary or idiopathic. If the seizures have a known cause, the condition is referred to as secondary or symptomatic epilepsy. Some of the more common causes include the following:
Before a patient can be considered for surgical treatment he has to undergo extensive preoperative evaluation.
The goals for Epilepsy surgery are:
The main aim of presurgical evaluation in patients with intractable epilepsy is the identification of the cortical area capable of generating seizures and whose removal or disconnection will result in seizure freedom. This area is called the epileptogenic zone. Different diagnostic tools are being used by epileptologist to identify epileptogenic zone.
The current diagnostic techniques used in the definition of these cortical zones for epilepsy surgery are video electroencephalography (EEG) monitoring, magnetic resonance imaging (MRI), ictal single photon emission computerized tomography (SPECT) and positron emission tomography (PET). A detailed neuropsychological evaluation is an indispensable tool for prognosis of neuropsychological deficits after surgery and may significantly influence the final decision about epilepsy surgery.
Magnetic resonance imaging
The common abnormalities identified by MRI in patients with refractory epilepsy are mesial temporal atrophy and sclerosis (MTS), malformations of cortical development, primary brain tumors, vascular malformations and focal atrophic lesions. MRI have to be performed using epilepsy protocol to ensure maximal yield in picking up the abnormalities. Sometimes the presurgical evaluation team may ask for a repeat MRI if they do not agree with the MRI findings. MRI remains one of the strong tools in defining the candidature for surgery.
Other than MTS, malformations of cortical development are being increasingly recognized in patients with refractory epilepsy. They may be focal cortical dysplasia (FCD), lissencephalies, heterotopia, polymicrogyria, schizencephaly. Patients with low-grade primary brain tumors frequently present with seizures. The underlying histopathologies include dysembryoplastic neuroepithelial tumors, ganglio-glioma, gangliocytoma and pilocytic and fibrillary astrocytoma. Newly developed MRI techniques, diffusion-weighted imaging (DWI), diffusion tensor imaging (DTI), tractography improve the sensitivity of MRI and help in surgical planning.
Functional Imaging
Ictal (during seizure) single photon emission computerized tomography (SPECT) and interictal (in-between seizures) positron emission tomography (PET) remain important imaging tools in the presurgical evaluation of patients with refractory partial epilepsy. SPECT measures blood flow; and comparing interictal and ictal SPECT studies, the increase in blood flow of certain brain regions during the ictal phase with respect to the interictal period can be evaluated. During ictal SPECT, due to epileptic activation, the neurons located in these areas are hyperactive and there is an increase in blood flow as an autoregulatory response. An ictal SPECT displays both the ictal onset zone and seizure propagation pathways. In common practice, the region with largest and most intense hyper-fusion is considered as the ictal onset zone.
18 F-deoxyglucose (FDG) PET measures changes in cerebral glucose metabolism and has higher spatial resolution and more reliable quantitation than SPECT, but the temporal resolution of PET with 18 FDG is unfavourable for ictal studies. PET maps cerebral glucose metabolism using FDG PET and cerebral blood flow using 15 O-labelled water. Regional hypo-metabolism is best analysed with co-registration of PET scans to MR images. The sensitivity of FDG PET is 60-90% for the detection of interictal temporal lobe hypo-metabolism. FDG PET is more useful for lateralizing than localizing the epileptic focus. Patients with MTS have low glucose metabolism in the whole temporal lobe while patients with mesiobasal temporal tumors show only a slight decrease in metabolism.
Functional magnetic resonance imaging
Functional MRI (fMRI) helps to visualize regional brain activity. It provides a reliable way to lateralize language dominance and eliminates the need for invasive intra-carotidamobarbital test (IAT) in 80% or more patients. A series of related tests, such as verbal fluency and language comprehension, are administered during MRI to localise the speech centre. Similarly, in patients with lesions near the motor area (area responsible for power) the relation of the lesion to the motor strip can be defined using fmri. This helps in surgical planning of the excision of lesion.
Role of EEG in Presurgical Evaluation
Non-invasive EEG monitoring (Video telemetry)
Long-term non-invasive video EEG monitoring in presurgical evaluation is performed to differentiate seizure versus non seizure events, classification of seizure types and localization of seizure onset. It is expensive and labour intensive. At least two to five habitual seizures should be recorded after gradual AED withdrawal. The aim is to pin point the onset of the electrical seizure and correlate it with the seizure manifestation (semiology). At Jaslok hospital we perform this in ICU to avoid any untoward complications.
In cases where we cannot conclude from non-invasive EEG we would perform invasive EEG. This are in the form of subdural grids and depth electrode recordings.
Neuropsychological evaluation
The primary goal for neuropsychological evaluation is to characterize the patient’s intellectual level, intelligence quotient (IQ) with the Wechsler Adult Intelligence Scale (WAIS) or a revision of it (WAIS-R), the Minnesota Multiphasic Personality Inventory (MMPI) and the Washington Psychosocial Seizure Inventory (WPSI). An epileptic dysfunction in a silent cortical area will have less influence on IQ area. Discriminative neuropsychology has several tests in store. Neuropsychology provides information about size, location and degree of epileptic dysfunction. Preoperative evaluation assists in predicting epilepsy surgery outcome and thus helps in selecting ideal candidates for surgery. Epilepsy surgery can be performed without any neuropsychology at all, but it helps in the preoperative counselling of the patients and their caregivers. It provides baseline values against which the postoperative values can be compared.
Successful epilepsy surgery in India requires a multidisciplinary team approach with discussion of individual patient presurgical evaluation data in detail in a patient management conference. It will improve patient care and communication among members of the team.
There are several epilepsy surgery technologies but the most common epilepsy surgery is Temporal Lobe epilepsy surgery. Medial temporal sclerosis (MTS) is one of the best-characterized epileptic syndromes. MTS is defined by classical temporal lobe seizures, associated with characteristic temporal spikes on EEG and hippocampal sclerosis identified on MRI. The improvement in seizure frequency after temporal lobectomy is measured by reduced frequency and intensity. Based on the Engel’s classifications, improvement ranges from 60% to 80%. In select populations, seizure-free outcomes of 90% to 100% have been reported.

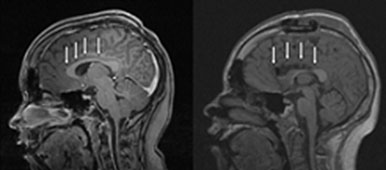
MRI Showing Mesial Temporal Sclerosis
Furthermore, neuropsychologic outcomes are improved after temporal lobectomy. In one study, an average 10-point gain in IQ was seen in children after temporal lobectomy. The best outcomes with respect to neurocognitive function are seen when surgery is performed during adolescence or even earlier. In a double blind study performed on adult patients suffering from MTS it was concluded that surgical treatment was superior to prolonged medical therapy. In our personal series of over 100 cases there has been no mortality or major permanent morbidity. The seizure outcome (Engel gr I & II, i.e. almost no seizures) has been 77%.

Picture after the resection of Temporal Lobe Epilepsy Surgery
This outcome is the best possible epilepsy surgery outcome amongst all epilepsy surgery.
Various techniques are used for temporal lobectomy. At JHRC we perform what is called standard temporal lobectomy with hippocampal resection in all cases.
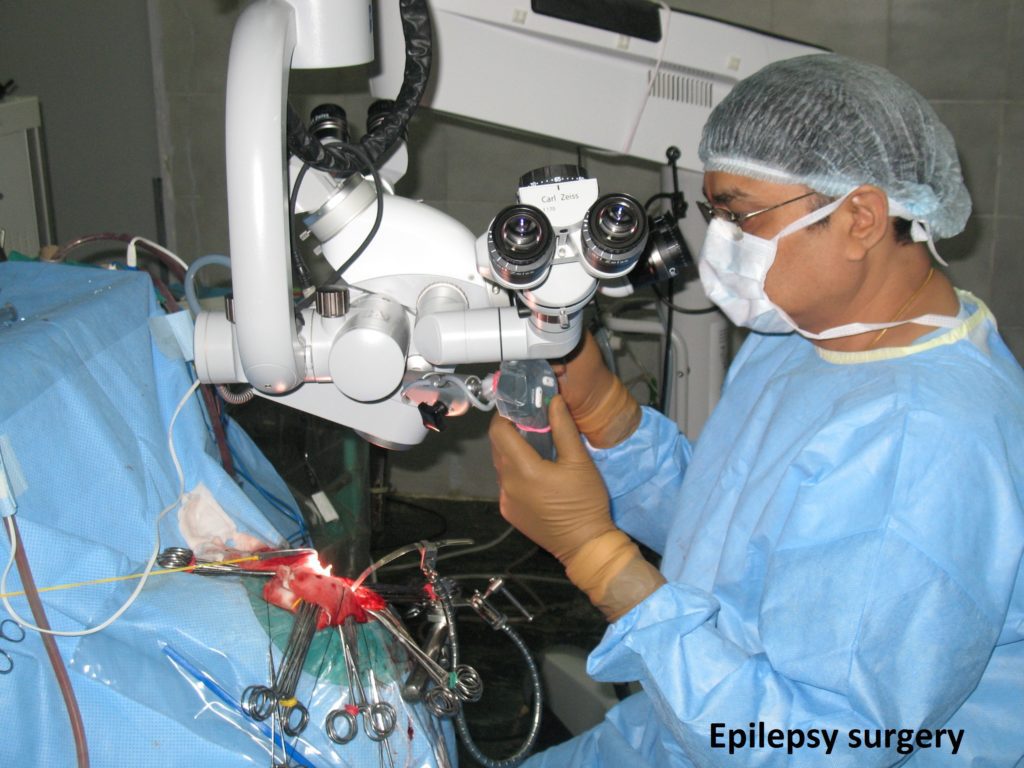
Removal/Disconnection of malfunctioning one half of the brain is known as hemispherectomy/hemispherotomy. McKenzi is credited to perform the first hemispherectomy to treat intractable epilepsy on an adult patient in 1938 with excellent results. The procedure gained popularity in the following decades with excellent results and acceptable mortality rates.
Rasmussen suggested modifications by removing only certain portions and disconnecting the remaining cortex, which was left in situ. The goal was to reduce the subdural cavity, thus reducing the risk of hemorrhages. Various modifications have been suggested aimed at preservation of the cortex with disconnection, hence reducing the risk of complications.
Indications
The typical applicants are patients with stubborn epilepsy due to autonomous hemispheric cerebral pathology. These include congenital malformations such as Sturge-Weber syndrome, Hemimegalencephaly, diffuse cortical dysplasia or acquired lesions such as Rasmussen encephlalitis. However, the most common pathology is perinatal infarct.
At Jaslok hospital, detailed clinical evaluation with documentation of hemiparesis and hemianopsiaperformed. Bilateral pathology or deficits predicts poor seizure-free outcome. Detailed imaging and electrophysiology will help predict the outcome. In certain cases where bilateral pathology is noted, improvement may still be noted in patients who undergo the procedure on the worst side. From the standpoint of producing a neurologic deficit, it is often already achieved by the disease process. In a progressive condition, such as progressive chronic encephalitis (Rasmussen’s), extensive Sturge-Weber syndrome and infantile spasms, in which continued clinical deterioration leading to a maximum contralateral neurological deficit is expected, early hemispherectomy may cause an acute worsening of the neurological deficit, but not worse than that to be produced by the disease process. The improved seizure control and psychosocial improvement following successful surgery outweigh the poor prognosis associated with the natural history of these disease processes, which are progressive and uniformly refractory to medical therapy.
Timing of Epilepsy Surgery
If proof of unilateral hemispheric damage is found, then the timing is determined by the severity and the age of the patient. Hemispheric procedure in the second or third year of life does not carry any risk of increased deficit and hence ideal for patients who come at early stages for diagnosis and evaluation. In late onset cases such as Rasmussen’s encephalitis, timing is controversial; as language and motor function transfer is less likely in older children. Evidence is accumulating that seizures themselves as opposed to the pathological substrate may significantly delay the cognitive development. Timing of surgery, to a great extent, depends on the severity of the epilepsy, natural history of the disease, adequacy of therapeutic medical trial. In certain conditions which are inborn, such as hemimegalencephaly and diffuse cortical dysplasia, very early surgery may be optimal.
Surgical Techniques
The surgical techniques of hemispherectomy have been historically classified into two main categories: the classic ‘anatomic hemispherectomy’ with its variations and the functional hemispherectomy. The anatomic hemispherectomy has had multiple modifications to decrease the complication rate, more specifically from superficial cerebral hemosiderosis and hydrocephalus. The functional hemispherectomy, initially described by Rasmussen and more recently modified by Villemure, minimizes the resection of tissue and has been shown to likewise further reduce the long-term complications.
The anatomic hemispherectomy simply consists of the removal of the cerebral hemisphere without the basal ganglia. The procedure may be done either en bloc or in multiple lobectomies, depending on the surgeon’s preference. Complications encountered following this method of hemispherectomy have led many centres to abandon it in its original form and have attempted multiple modifications.
‘Functional hemispherectomy’ is more widely used presently and consists of an anatomically subtotal, but physiologically complete, hemispherectomy. The operation is based on principles of disconnection rather than excision. The originally described method requires the excision of the central frontoparietal cortex, including the parasagittal tissue from the level corresponding to the genu of the corpus callosum to the splenium; a temporal lobectomy with an excision of amygdala and hippocampus; disconnection of the residual frontal and parieto-occipital lobes medially; a corpus callosotomy and an insulectomy.
Complications
A frequent postoperative complication following hemispherectomies is persistently elevated systemic temperature for 8-12 days postoperatively, which is likely to be secondary to the blood products entering the CSF. Early postoperative brain shift, with herniation and death, has been reported following anatomic hemispherectomy. Hydrocephalus in the early postoperative period, not related to superficial cerebral hemosiderosis has been reported in 7.5-10.5% of patients.
Late postoperative complications include infections and spontaneous and posttraumatic hemorrhages. The hemispherectomy-associated mortality was reported in a range between 1 and 6.6% however; in the recent series, this is closer to the lower end. Mortality is usually related to uncontrollable intraoperative blood loss and with the development of obstructive hydrocephalus.
At the Jaslok Hospital, we perform hemispherotomy in all patients with hemispheric dysfunction. In a carefully selected patient population, the seizure freedom has been almost 100%. We have had no operative or perioperative deaths. There has not been a single case of postoperative increase in the neurological deficit.
Stimulation of the central nervous system (CNS) is a novel technique under evaluation for medically intractable epilepsy. CNS stimulation for epilepsy has been a matter of extensive research. Cerebellar stimulation has been reported to reduce seizures. Vagal nerve stimulation (VNS) has been approved by US FDA for use in epilepsy since 1997. Direct cortical stimulation, which was previously used as a diagnostic tool, now finds a place in therapeutics. Another modality of electrical stimulation being studied in research settings is repetitive transcranial magnetic stimulation (rTMS), which is the simplest and least invasive approach. Various institutions are also designing clinical trials to study the therapeutic effects of deep brain stimulation (DBS), either targeting the subthalamic or anterior thalamic nucleus, in the management of epilepsy. While preliminary studies regarding stimulation in the treatment of epilepsy have been suggestive, double-blind studies with larger number of patients will be needed before any definitive answers are found. The role of subacute hippocampal electrical stimulation (SAHCS) is also being evaluated.
Neurophysiological Basis
The CNS is primarily a neural network. Neurostimulation is a means to modulate the information-processing activity of the CNS, so as to correct electrical dysfunction. This is typically carried out to compensate for the loss of normal function. It is important to localize specifically what part of the circuit has to be targeted and which is the part that is malfunctioning.
Vagal nerve stimulation (VNS)
As of today, VNS is an approved therapy for the management of medically intractable partial-onset epilepsy. VNS therapy is indicated for use as an adjunctive therapy in reducing the frequency of seizures in patients whose epileptic disorder is dominated by partial seizures (with or without secondary generalization); or generalized seizures, which are refractory to antiepileptic medications.
Till mid-2006, about 40,000 people had been treated with VNS. About 80% of all implanted patients have some seizure improvement while on VNS therapy and more than 40% of all implanted patients have a greater than or equal to 50% reduction in the number of seizures over time. It has also been found that the antiepileptic effects of VNS improve over time. Zabara first reported vagal stimulation as a modality of treatment for epilepsy, Woodbury confirmed his results in the rat model. A randomized control trial of 125 patients showed a reduction in seizure frequency by 24.5%, using high-stimulation parameters. The first VNS implantation in a human patient was done in November 1998.
VNS is indicated for use as an adjunctive therapy in reducing the frequency of seizures in patients whose epileptic disorder is dominated by partial seizures (with or without secondary generalization); or generalized seizures, which are refractory to antiepileptic medications. It can also be used in patients who have medical contraindications for surgery.
A preoperative evaluation includes video-EEG, magnetic resonance imaging (MRI), positron emission tomography (PET) and evaluation by a multidisciplinary team.
Technical aspects
Left vagus nerve is selected for stimulation. It is approached through a carotid or transverse neck incision at the mid-neck level. The main vagal trunk is identified and exposed for 3-4 cm in the carotid sheath. Electrode coils are passed around the nerve without putting undue tension on the nerve or the coil. The electrodes are tunneled subcutaneously and connected to a pacemaker (after trial stimulation) implanted in the infraclavicular region.
Labar et al. have found a decline in seizure rate by 37% at one year and 43% at two and three years. Menachem et al. also report an average seizure rate reduction of 43%. VNS has been shown to benefit primary generalized seizures as well. Adverse effects include coughing, hoarseness (most common), dyspnea, vocal cord paralysis, infection, Horner’s facial palsy, hardware-related complications and death. Contraindications include patients with a history of prior left neck surgery or vagal surgery.
Anterior thalamic nucleus stimulation
Recently, deep brain stimulation (DBS) of the anterior thalamic nucleus (AN) has come up again as treatment for medically intractable seizures. The therapy has its basis in studies done in the 1940s and 1950s. It was introduced in human subjects by Cooper in the 1970s. Rationale: Although its mechanism is not well understood, DBS produces a functional lesion in the brain likely through depolarization blockade. It has been found that a specific subcortical pathway that synaptically links the anterior thalamic nuclear complex (AN) to the hypothalamus and midbrain is important in the expression of pentylenetetrazole (PTZ) seizures. Disturbance of neuronal activity along this path via focal disruption or chemical inhibition significantly raises seizure threshold. It was also seen that high-frequency (100 Hz) stimulation of AN did not alter the expression of low-dose PTZ-induced cortical bursting but did raise the clonic seizure threshold compared to naive animals or those stimulated at sites near, but not in, AN. The basic procedure is the same as for DBS for Parkinson’s disease. A superior frontal approach is used to approach the AN. Stereotactic targeting and MRI visualization are used as a guide to the target, i.e., the dorsal anterior portion of the thalami.
A study done at St. Joseph’s Hospital and Medical Centre, Phoenix, Arizona, found that in four of their five patients, stimulation showed clinically and statistically significant improvement with respect to the severity of their seizures, specifically with respect to the frequency of secondarily generalized tonic-clonic seizures and complex partial seizures associated with falls. No adverse events could clearly be attributed to stimulation. A further study by Hodaie et al. at the University of Toronto used AN stimulation in five patients suffering from medically intractable epilepsy. They found a statistically significant decrease in seizure frequency, with a mean reduction of 54% (mean follow-up, 15 months). Two of the patients had a seizure reduction of more than or equal to 75%. No adverse effects were observed after DBS electrode insertion or stimulation. The observed benefits did not differ between stimulation-on and stimulation-off periods.
Subthalamic nucleus (STN) stimulation
In carefully selected patients, STN stimulation is an accepted modality of management of Parkinson’s disease. It was the Grenoble group that initially described chronic electrical stimulation of this region. The first patient was reported in 1994; later they described the first DBS for Parkinson’s disease. Recently, chronic STN stimulation has been sought as a potential treatment for medically intractable epilepsy. Rationale: The subthalamic nucleus exerts an excitatory control on the nigral system. It has been seen that pharmacological or electrical inhibition of the STN leads to suppression of attacks in animal models of epilepsy. Benabid et al. Performed STN high-frequency stimulation (HFS) in five patients suffering from medically intractable seizures and considered unsuitable for respective surgery. A 67% to 80% reduction in seizure frequency was observed in three patients, with partial symptomatic epilepsy of the central region. An additional patient suffering from severe myoclonic epilepsy also responded to STN HFS, with a weaker reduction of seizure frequency. The Cleveland study in patients with medically intractable nonsurgical focus epilepsy found that high-frequency stimulation of the subthalamic nucleus (STN) was effective in two of its four patients, with a marked decrease in seizure frequency – ranging from 42% to 75%. Constant and intermittent stimulation modes were similarly effective. These two patients also reported that STN stimulation elicited a significant decrease in seizure severity and duration.
Conclusion
Intractable epilepsy forms 30% of all treated patients. Surgery cannot be offered to all such patients. Thus arises the need for alternative modalities of treatment. VNS has shown very good results and has been approved by US FDA. TMS and TCS have also shown promising results. Deep brain stimulation (hippocampal, anterior thalamic and STN) and RNS are newer modalities under evaluation.
The first examples of corpus callosotomy were performed in the 1940s by Dr. William P. van Wagenen; the corpus callosum is a fibre bundle of about 300 million fibrein the human brain that connects the two cerebral hemispheres. The interhemispheric functions of the corpus callosum include the integration of perceptual, cognitive, learned, and volitional information.
The role of the corpus callosum in epilepsy is the interhemispheric transmission of epileptiform discharges. These discharges are generally bilaterally synchronous in preoperative patients. In addition to disrupting this synchrony, corpus callosotomy decreases the frequency and amplitude of the epileptiform discharges, suggesting the transhemispheric facilitation of seizure mechanisms. Seizure surgery involves:
A corpus callosotomy is an operation that severs (cuts) the corpus callosum, interrupting the spread of seizures from hemisphere to hemisphere. Seizures generally do not completely stop after this procedure (they continue on the side of the brain in which they originate). However, the seizures usually become less severe, as they cannot spread to the opposite side of the brain.
Indications
Patients of any age, including infants, may be considered for surgery if other criteria are met.
Contraindications
Decision making
Preoperative evaluation
Note
Surgical procedure: CORPUS CALLOSUM SECTION (CORPUS CALLOSTOMY)
A corpus callosotomy requires exposing the brain using a procedure called a craniotomy. After the patient is put to sleep with anaesthesia, the surgeon makes an incision in the scalp, removes a piece of bone and pulls back a section of the dura, the tough membrane that covers the brain. This creates a “window” in which the surgeon inserts special instruments for disconnecting the corpus callosum. The surgeon gently separates the hemispheres to access the corpus callosum. Surgical microscopes are used to give the surgeon a magnified view of brain structures.
In some cases, a corpus callosotomy is done in two stages. In the first operation, the front two-thirds of the structure is cut, but the back section is preserved. This allows the hemispheres to continue sharing visual information. If this does not control the serious seizures, the remainder of the corpus callosum can be cut in a second operation. After the corpus callosum is cut, the dura and bone are fixed back into place, and the scalp is closed using stitches or staples. After the operation, most patients are monitored in an intensive care unit for 24 to 48 hours.
The patient generally stays in the hospital for 6-10 days. Most people having a corpus callosotomy will be able to return to their normal activities, including work or school, in six to eight weeks after surgery. The hair over the incision will grow back and hide the surgical scar. The person will continue taking anti-seizure drugs.
After the surgery, the patient may experience temporary or permanent limitations with speech, behavior, or movement of certain body parts. It is important for patients and families to understand that these risks exist. This surgery is not done to cure the seizures, but rather to help reduce their severity.
Outcome and prognosis
The following symptoms may occur after having a corpus callosotomy, although they generally go away on their own:
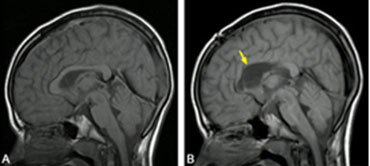
Risks of a Corpus Callosotomy
The most prominent non-surgical complications of corpus callosotomy relate to speech irregularities. For some patients, sectioning may be followed by a brief spell of mutism. A long-term side effect that some patients may suffer is an inability to engage in spontaneous speech. In addition, the resultant split-brain prevented some patients from following verbal commands that required used of their non-dominant hand.
Surgery is indicated in patients where more than two drugs for more than two years are tried, and still the disease is not adequately controlled. 15-20% of all epilepsy patients require surgery.
The epileptogenic area is either resected (temporal lobe resection in Mesial temporal sclerosis), any low grade tumour or disconnected callosotomy, hemispherotomy, subpial transection.
he relief is complete in 67-70% of temporal lobe epilepsy and 60-65% of extra-temporal epilepsy.
The drugs are tapered over the period of one year with the help of epileptologist.
The complications of epilepsy surgery depend upon the nature of the surgery. They can range from motor weakness, speech problems or some memory disturbances. However, in a carefully selected patient and properly performed epilepsy surgery the complication rate can be less than 2%.
Average hospital stay is 7-10 days
WhatsApp us